The effects of stimulus contrast on visual perception have been known for a long time. For example, there is a consensus that at lower contrasts objects appear to be moving slower than they actually are (Thompson, 1982). Several computational models have been posited to account for this observation, although up until now there has been a paucity of neural data with which to validate them. A recent article in the Journal of Neuroscience seeks to address this issue, combining single-cell recordings in a motion sensitive region of visual cortex with psychophysics and modeling to better elucidate the neural code for speed perception (Krekelberg et al, 2006).
It has been well established that the middle temporal (MT) cortical area in the macaque monkey is of central importance for both motion processing and perception (for review, see Born and Bradley, 2005). Importantly, it has recently been shown that lowering stimulus contrast produces a qualitative difference in the extent to which MT cells integrate information over space (Pack et al, 2005). Hence, it is only natural to wonder how the joint manipulation of stimulus contrast and speed affects both perceptual reports and MT responses.
To answer this question, Krekelberg et al. recorded neural activity from single units in area MT of awake-behaving macaques in response to patches of moving dots that could vary both in terms of speed and contrast. The purpose of the electrophysiological recordings was to elicit neural speed tuning curves at various levels of contrast. In these trials, experimenters presented a single patch that was centered on the receptive field and moving in the preferred direction of the neuron while the monkey maintained fixation. In separate sessions, the authors had human observers and a monkey subject perform a psychophysical speed discrimination task, thus allowing them to compare neural and psychophysical performance. In the psychophysical task, observers had to judge which of two simultaneously presented patches of moving dots appeared faster – one patch was presented at a fixed speed but variable levels of contrast while the other was presented at a fixed contrast level but at a variable speeds. This procedure yielded psychometric functions quantifying the shift in apparent speed at lower contrasts.
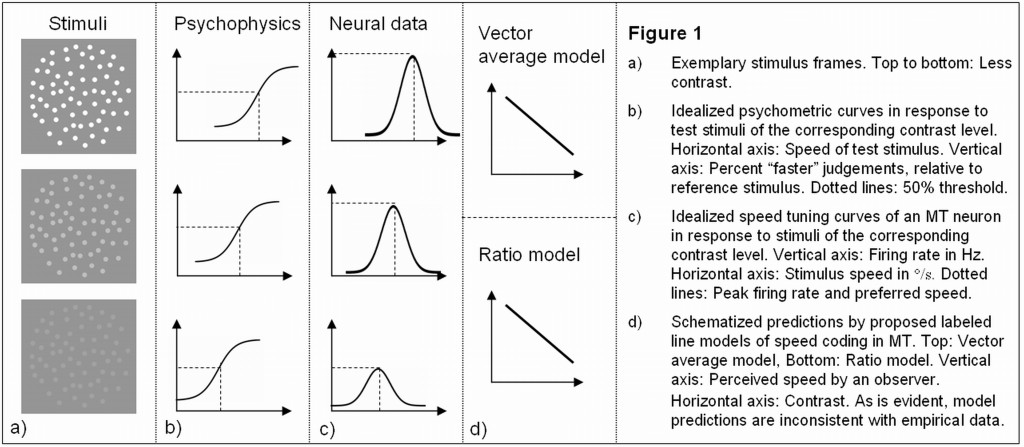
Figure 1
Using these methods, the authors report several major findings.
Consistent with previous reports (Thompson 1982), the authors show that there is a substantial effect of contrast on speed perception, insofar as the perceived speed of the random dot stimuli is drastically reduced at low contrasts – reducing contrast by one octave reduces the perceived speed by about 9%. Moreover, the psychophysical data from the monkey suggest that there is – at least qualitatively – a corresponding effect in macaques.( See Krekelberg et al., Figure 2).
Further, reducing contrast had several effects on the neural activity of speed tuned MT neurons: Generally, speed tuning curves shift such that the peak firing rate is reached at slower speeds (Krekelberg et al., Figure 5A, 6A) – this shift in preferred speed is more pronounced in neurons preferring faster speeds (Figure 5B). Also, most – but not all – cells respond less vigorously at lower contrasts (Krekelberg et al., Figure 4A). It is of particular interest to note that due to the shifted peak, some cells (about 30% of neurons) respond more strongly in response to low contrast than to high contrast motion at slow speeds. (See Krekelberg et al., Figure 3C).
Finally, the authors use these neural data to test models attempting to account for the observed psychophysical effects. Specifically, they test two exemplars of a family of labeled line models, each of which had previously been shown to account for human speed perception. In the vector average model, a population of MT cells effectively computes a weighted average, where each neuron’s contribution is proportional to its preferred speed and a weight given by its normalized firing rate. Surprisingly, when fed with their neural data, the authors find that this model predicts an increase in perceived speed with lower contrast, inconsistent with the psychophysical data, (Krekelberg et al., Figure 7).
Similarly, the authors point out that a “ratio model”, in which perceived speed corresponds to the ratio of activity in a fast and a slow channel, also cannot account for the psychophysical effects in terms of the neural data (Krekelberg et al., Figure 8B).
Hence, the authors state that their data is fundamentally inconsistent with existing labeled-line models of speed coding in area MT. They conclude that it is likely that the code for speed in MT will differ from the established labeled-line codes that can account for direction perception.
While there are several potential problems which will need to be addressed in further work, specifically: stimulus size was held constant (potentially covering a dynamic surround), not only contrast but also luminance varied across conditions, and finally, there was a profound lack of psychophysical data in the macaque, insofar as we can tell it appears that none of these issues threatens the interpretations proffered by the authors.
The authors characterize their effect as the result of imperfect contrast-gain control and to a large degree, the differences in firing rates at different contrasts can be accounted for by simple contrast gain mechanisms (Krekelberg et al, Figure 4C). However, before one can attribute the observed effects to such a mechanism, it is beneficial to first rule out other plausible explanations.
A more exciting view, highlighting the functional role of non-veridical speed perception has recently been proposed. In this Bayesian approach, it is adaptive, when signal strength is low in proportion to the noise, to rely less on current sensory input and more on prior experience, which recent evidence suggests corresponds to slower speeds (Stocker & Simoncelli, 2006).
Of course, this doesn’t detract from the theoretical significance, or the main thrust of the paper, which is highly provocative as it highlights deficits in current models of speed perception. To improve on these models, it might be beneficial to simultaneously gather neural and psychophysical data from animals performing a similar speed discrimination task. This would allow a more direct comparison of neurometric and psychometric performance measures as well as the elucidation of neuro-perceptual correlates. Additionally it would provide modelers with valuable data on the issue of speed coding in area MT.
In conclusion we believe that this paper proposes a formidable challenge to both the neurophysiological as well as the modeling community.
References
Born, R. T. & Bradley, D. C. (2005) Structure and function of visual area MT. Annu. Rev. Neurosci., 157-189.
Krekelberg, B., van Wezel, R.J.A., & Albright, T.D. (2006). Interactions between Speed and Contrast Tuning in the Middle Temporal Area: Implications for the Neural code for Speed. J. Neurosci., 8988-8998.
Pack, C. C., Hunter, J. N., & Born, R. T. (2005) Contrast dependence of suppressive influences in cortical area MT of alert macaque. J. Neurophysiol., 1809-1815.
Stocker, A. A. & Simoncelli E. P. (2006). Noise characteristics and prior expectations in human visual speed perception. Nat. Neurosci., 578-585.
Thompson, P. (1982) Perceived rate of movement depends on contrast. Vision Res., 377–380.
Acknowledgments: This piece was written with substantial input from Andrew M. Clark